
It was met with skepticism but later proved correct when X-ray crystallography confirmed it about the keratin protein’s structure. Linus Pauling proposed the model for alpha helical structures in proteins in 1951. It also plays a role in various protein functions like membrane anchoring and recognition. Side chain orientation: Chains project outward from the helical axis.Īlpha helix is vital for efficient packing of amino acids, reducing steric clashes and maximizing protein stability. Residue arrangement: Amino acid residues along the same axis.ĭipole moment: Parallel to helical axis – important for protein-protein interactions. It has specific characteristics that give it stability and function.īackbone arrangement: Hydrogen bonds between peptide bonds for stability. Definition and characteristics of alpha helixĪlpha helix is a right-handed coiled structure found in proteins. The alpha helix plays a crucial role in determining a protein’s folding, stability, and functionality. We’ll then explore how this specific secondary structure holds great importance in the overall structure of proteins. To understand the alpha helix, let’s dive into its definition and characteristics. Q: What is the Helical Wheel in an Alpha Helix?.Q: What is the Helix Axis in an Alpha Helix?.Q: What is the N-terminus of an Alpha Helix?.Q: What is the sidechain in an Alpha Helix?. 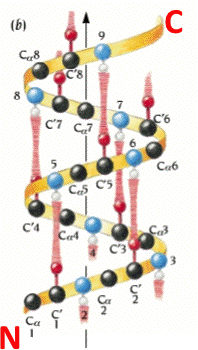
Q: What is the primary structure of an Alpha Helix?.Q: What are the functions of an Alpha Helix?.Q: What is the structure of an Alpha Helix?.Q: What are the characteristics of an Alpha Helix?.Relationship between the alpha helix and three-dimensional protein structure.Alpha helix bundles and their role in protein folding.Alpha helix in tertiary protein structure.Coiled-coil and leucine zipper structures formed by alpha helices.Left-handed alpha helix and its occurrence.Variations and modifications of alpha helix.Nuclear magnetic resonance (NMR) spectroscopy in studying alpha helices.X-ray crystallography and alpha helix visualization.Experimental Evidence for alpha helix structure.Functional motifs formed by alpha helices.Role of alpha helix in membrane-spanning proteins.Influence of the amino acid side chains on alpha helix stability.Role of proline in disrupting alpha helices.Factors affecting alpha helix formation.Propensity of amino acids to form alpha helices.Secondary structure elements in alpha helix.Influence of amino acid residues on alpha helix stability.Role of hydrogen bonding in alpha helix formation.Importance of alpha helix in protein structure.Definition and characteristics of alpha helix.Modern techniques for circular dichroism spectroscopy. Cambridge: Royal Society of Chemistry 2010. Linear dichroism and circular dichroism: a textbook on polarized spectroscopy. An Escherichia coli twin-arginine signal peptide switches between helical and unstructured conformations depending on hydrophobicity of the environment. Miguel MS, Marrington R, Rodger PM, Rodger A, Robinson C. Analyzing protein circular dichroism spectra for accurate secondary structures. Single peptide bonds exhibit poly(pro)II (“random coil”) circular dichroism spectra. The effect of conformation on the CD of interacting helices: a theoretical study of tropomyosin. Comprehensive chiroptical spectroscopy (in press).Ĭooper TM, Woody RW. A natively unfolded toxin domain uses its receptor as a folding template.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
